Structure-Based Virtual Screening Identifies Ritobegron and Resveratrol as Dual Inhibitors of NMDA and PTP-1B for Alzheimer’s Disease Therapy
Main Article Content
Abstract
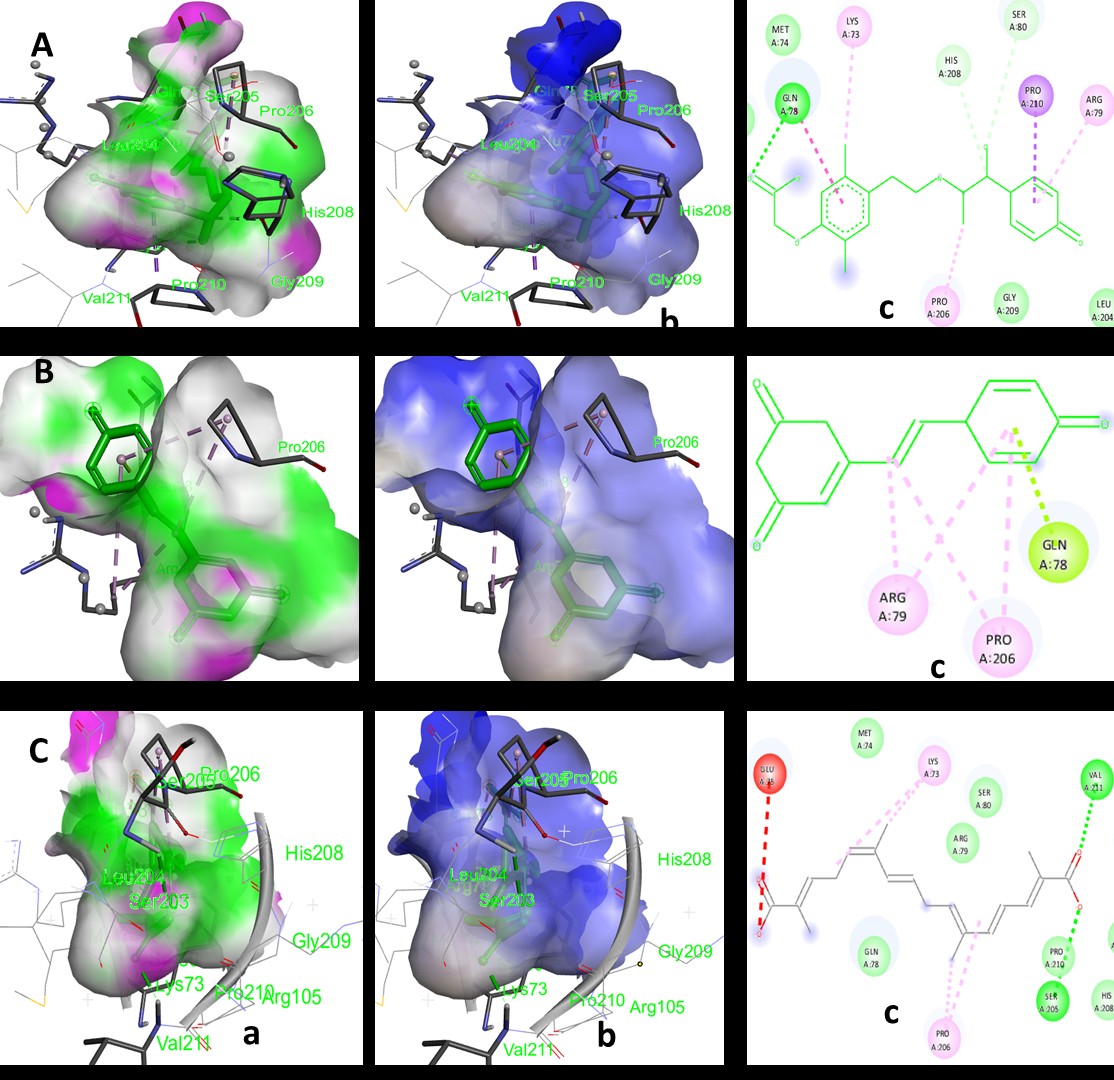
Many Alzheimer’s disease (AD) drug candidates fail during clinical translation due to poor target validation and inadequate penetration across the blood–brain barrier. This study aimed to identify potential inhibitors for AD by targeting N-methyl-D-aspartate (NMDA) and protein tyrosine phosphatase 1B (PTP1B). A total of 2,399,743 molecules retrieved from the ChEMBL database were filtered using various in silico techniques to identify small, investigational molecules under phase III clinical trial, that are safe, bioavailable with favourable physicochemical properties. The top-ranked molecules were subjected to molecular docking studies on NMDA and PTP1B. A total of 1,920,643 small molecules were identified, among which 1,110 investigational molecules had advanced to phase III clinical trials. Of these, 298 molecules met the Lipinski Rule of Five and were considered safe, and ultimately only 3 molecules demonstrated favourable physicochemical properties. Ritobegron, docked with PTP1B, showed a binding energy, E of -8.69 kcal/mol and an inhibition constant, Ki of 0.424 µM, outperforming the native ligand (-7.23 kcal/mol, 5.04 µM) and with NMDA showed E of -8.06 kcal/mol and Ki of 1.23 µM, both superior to its native ligand (-7.40 kcal/mol, 3.74 µM). Resveratrol, docked with protein PTP1B, showed E of –7.59 kcal/mol and Ki of 2.53 µM, slightly more negative than the native ligand (–7.23 kcal/mol, 5.04 µM). These interactions involved hydrogen bonding, pi-stacking, and hydrophobic interactions with key amino acid residues. The study suggests that ritobegron and resveratrol could serve as promising inhibitors for NMDA receptors and PTP1B enzyme, offering potential therapeutic avenues for AD treatment.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
1. Frisoni GB, Aho E, Brayne C, Ciccarelli O, Dubois B, Fox NC, Frederiksen KS, Gabay C, Garibotto V, Hofmarcher T, Jack CR. Alzheimer's disease outlook: controversies and future directions. The Lancet. 2025; 406(10510): 1424-1442. https://doi.org/10.1016/S0140-6736(25)01389-3.
2. Rahman A, Jaiswal A, Keshari P, Singh DK. Alzheimer’s: Epidemiology, pathophysiology, diagnosis, and treatments. In Proteostasis: Investigating molecular dynamics in neurodegenerative disorders 2025; 39-72. Singapore: Springer Nature Singapore. https://doi.org/10.1007/978-981-96-6202-9_2
3. Cummings J, Lee G, Nahed A, Kambar MEN, Zhong K, Fonseca J, Taghva K. Alzheimer’s disease drug development pipeline: 2022. Alzheimers Dement (NY). 2022; 8(1): e12295. https://doi.org/10.1002/trc2.12295
4. Scheltens P, De Strooper B, Kivipelto M, Holstege H, Chételat G, Teunissen CE, Cummings J, van der Flier WM. Alzheimer's disease. The Lancet. 2021; 397(10284): 1577-1590. https://doi.org/10.1016/s0140-6736(20)32205-4
5. Xing H, Yue S, Qin R, Du X, Wu Y, Zhangsun D, Luo S. Recent advances in drug development for Alzheimer’s disease: A Comprehensive Review. Int J Mol Sci. 2025; 26(8): 3905. https://doi.org/10.3390/ijms26083905
6. Hossain MS, Hussain MH. Multi‐target drug design in Alzheimer's disease treatment: emerging technologies, advantages, challenges, and limitations. Pharmacol Res Perspect. 2025; 13(4): e70131. https://doi.org/10.1002/prp2.70131
7. Zaki SB, Khan SA, Ali R. A multi-enzyme study reviewing the role of target enzymes in Alzheimer’s disease and unveiling potential inhibitors with insights on their present and future assessment. Med Chem Res. 2025; 34(3): 549-570. https://doi.org/10.1007/s00044-025-03373-w.
8. Kołodziej-Sobczak D, Sobczak Ł, Łączkowski KZ. Protein tyrosine phosphatase 1B (PTP1B): A comprehensive review of its role in pathogenesis of human diseases. Int J Mol Sci. 2024; 25(13): 7033. https://doi.org/10.3390/ijms25137033
9. Mendez D, Gaulton A, Bento AP, Chambers J, De Veij M, Félix E, Magariños MP, Mosquera JF, Mutowo P, Nowotka M, Gordillo-Marañón M. ChEMBL: towards direct deposition of bioassay data. Nucleic acids Res. 2019; 47(D1): D930-D940. https://doi.org/10.1093/nar/gky1075
10. Banerjee P, Eckert OA, Schrey AK, Preissner R. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2018; 46(W1); W257–W263. https://doi.org/10.1093/nar/gky318
11. Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE. The Protein Data Bank. Nucleic Acids Res. 2000; 28(1): 235–242. https://doi.org/10.1093/nar/28.1.235
12. Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J Comput Chem. 2009; 30(16): 2785-2791. https://doi.org/10.1002/jcc.21256
13. Adasme MF, Linnemann KL, Bolz SN, Kaiser F, Salentin S, Haupt VJ, Schroeder M. PLIP 2021: Expanding the scope of the protein–ligand interaction profiler to DNA and RNA. Nucleic Acids Res. 2021; 49(W1): W530–W534. https://doi.org/10.1093/nar/gkab294
14. Furukawa H, Gouaux E. Mechanisms of activation, inhibition and specificity: crystal structures of the NMDA receptor NR1 ligand‐binding core. The EMBO J. 2003; 22(12): 2873–2885. https://doi.org/10.1093/emboj/cdg303
15. Krishnan N, Krishnan K, Connors CR, Choy MS, Page R, Peti W, Van Aelst L, Shea SD, Tonks NK. PTP1B inhibition suggests a therapeutic strategy for Rett syndrome. The J Clin Investig. 2015; 125(8): 3163-3177. https://doi.org/10.1172/jci80323
16. Banda SH, Uzonwanne MU, Didigwu OK, Nnadi CO. Cancer stem cells as potential targets of phytotoxic
17. alkaloids from the Toxic Plants-Phytotoxins database. Trop J Phytochem Pharm Sci. 2025; 4(2): 68-72. https://doi.org/10.26538/tjpps/v4i2.6
18. Nnadi CO, Ozioko LU, Eneje GC, Onah CM, Obonga WO. In-vivo antitrypanosomal effect and in-silico prediction of chronic toxicity of N-methylholaphyllamine in rats. Trop J Pharm Res. 2020; 19(11): 2369-2375. https://doi.org/10.4314/tjpr.v19i11.18
19. Nnadi CO, Onuku RS, Ayoka TO, Okorie HN, Nwodo NJ. Chemical constituents of Combretum dolichopetalum: Characterisation, antitrypanosomal activities and molecular docking studies. Trop J Pharm Res. 2022; 21(4): 801-808. https://doi.org/10.4314/tjpr.v21i4.17
20. Nnadi CO, Ngwu SO, Ohagwu MBZ. In-vivo and in-silico evidence of antitrypanocidal activities of selected plants from the Asteraceae family against Trypanosoma brucei brucei. Bioint Res Appl Chem. 2023; 13(1): 30. https://doi.org/10.33263/BRIAC131.030
21. Ezema IF, Akwu VC, Didigwu OK, Ogbonna JE, Ugwu AC, Nnadi CO. Exploring different drug targets responsible for the inhibitory activity of N, N'-substituted diamine derivative in Leishmania. Eng Proc. 2023; 56(1): 178. https://doi.org/10.3390/ASEC2023-16264
22. Smith DA, Beaumont K, Maurer TS, Di L. Relevance of half-life in drug design: Miniperspective. J Med Chem. 2017; 61(10): 4273-4282. https://doi.org/10.1021/acs.jmedchem.7b00969
23. Cottart CH, Nivet-Antoine V, Laguillier-Morizot C, Beaudeux JL. Resveratrol bioavailability and toxicity in humans. Mol Nutr Food Res, 2010; 54(1): 7–16. https://doi.org/10.1002/mnfr.200900437
24. Banks WA. From blood–brain barrier to blood–brain interface: New opportunities for CNS drug delivery. Nature Rev Drug Discov. 2016; 15(4): 275–292. https://doi.org/10.1038/nrd.2015.21
25. Abe Y, Nakano Y, Kanazawa T, Furihata T, Endo T, Kobayashi M. Investigation of drug–drug interactions between ritobegron, a selective β3‐adrenoceptor agonist, with probenecid in healthy men. Clin Pharmacol Drug Dev. 2016; 5(3): 201-207. https://doi.org/10.1002/cpdd.212.
26. Agnihotry S, Pathak RK, Srivastav A, Shukla PK, Gautam B. Molecular docking and structure-based drug design. In Computer-aided drug design 2020; 115-131. Singapore: Springer Singapore. https://doi.org/10.1007/978-981-15-6815-2_6
27. Podkowa K, Czarnacki K, Borończyk A, Borończyk M, Paprocka J. The NMDA receptor antagonists memantine and ketamine as anti-migraine agents. Naunyn-Schmiedeberg's Archiv Pharmacol. 2023; 396(7): 1371-1398. https://doi.org/10.1007/s00210-023-02444-2
28. Ye S, Han Y, Wei Z, Li J. Binding Affinity and Mechanisms of Potential Antidepressants Targeting Human NMDA Receptors. Molecules. 2023; 28(11):4346. https://doi.org/10.3390/molecules28114346